Delectrik’s products are based on vanadium redox chemistry and falls into the general class of redox flow batteries. This class of battery employs an electrolyte where energy is stored and a cell stack where energy conversion occurs. Energy is stored chemically in different ionic forms of vanadium in a dilute acid electrolyte. The electrolyte is pumped from separate plastic storage tanks into flow cells across a membrane where one form of electrolyte is electrochemically oxidized, and the other is electrochemically reduced. This creates a current that is collected by electrodes and made available to an external circuit. The reaction is reversible allowing the battery to be charged and discharged.
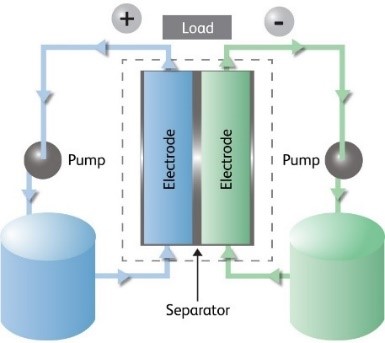
The redox flow battery is made up of two reservoirs to house the two different electrolyte solutions, and a “stack” of cells. Each cell has two half-cells, separated by a membrane and two current-collecting electrodes. One of the two different ionic forms of the electrolyte is in each half-cell. The positive and negative half-cells contain the electrolyte as Vanadium (IV/V) and Vanadium (II/III) redox couples, respectively. A pump supplies electrolyte to each half-cell, in a closed loop with the half-cell reservoir.
In the RFB the cells in the stack are electrically connected in series to build the stack voltage and hydraulic connection by the electrolyte is in a parallel fashion. The current density is determined by the surface area of the current collectors within the cell, but the supply of current depends on the electrolyte flow through the cells, and not on the stack itself.
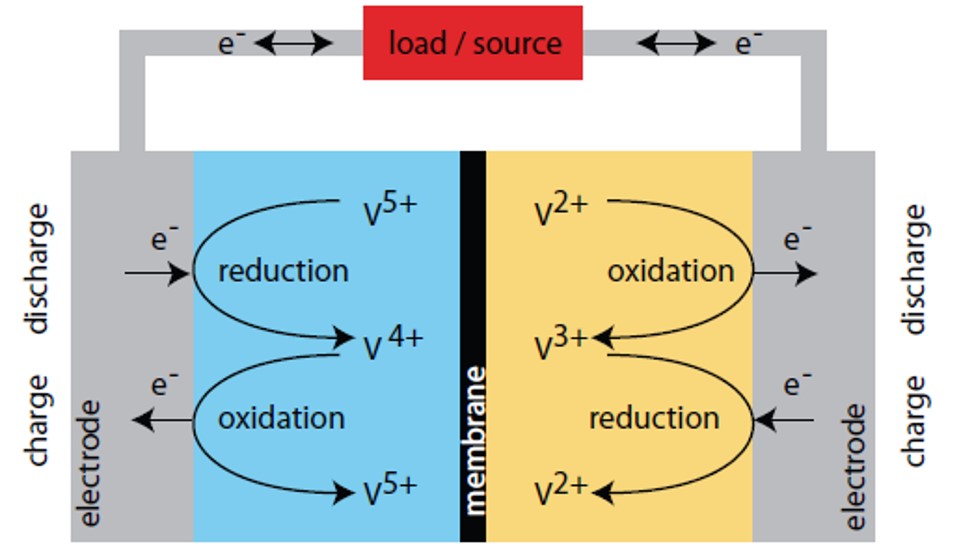
The RFB exploits the ability of vanadium to exist in 4 different oxidation states; the vanadium ions V4+ and V5+ are in fact vanadium oxide ions (respectively VO2+ and VO2+). Thus, the electrochemical equations become as follows.
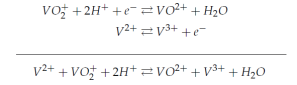
Where the water (H2O) and protons (H+) are required in the cathodic reaction to maintain the charge balance and the stoichiometry.
